TREMFYA® is the ONLY* IL-23i proven to significantly inhibit structural damage progression1-3†
IL-23=interleukin-23; IL-23i=interleukin-23 inhibitor; LAAD=Longitudinal Access and Adjudication Data; PsA=psoriatic arthritis.
*Based on approved IL-23 inhibitors for active PsA as of November 2025.
Source: IQVIA Weekly LAAD as of November 2025 X Free goods; Total PsA EqU share for Rheumatologists.
†Major secondary endpoint in a phase 3b study.
IN ADULT PATIENTS WITH ACTIVE PsA
APEX was conducted to build upon the DISCOVER program, which showed a reduction in structural damage progression but was not statistically significant4,5
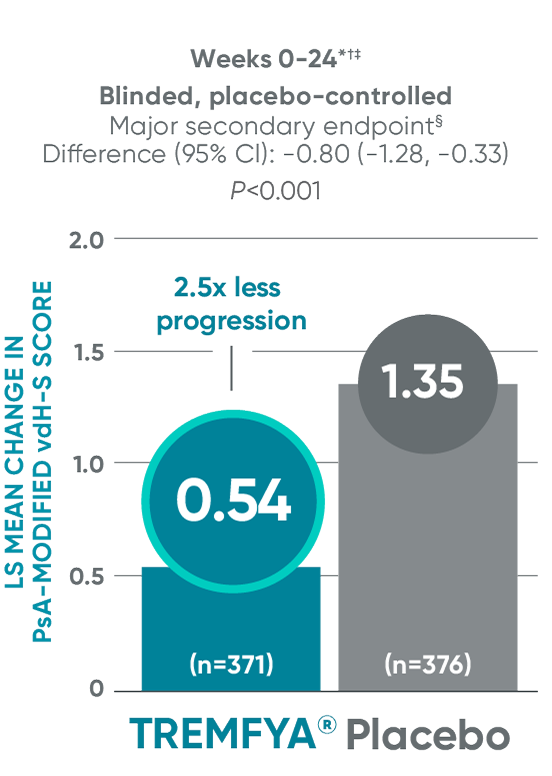
DISCOVER 2: Change from baseline in PsA-mvdH-S score: Blinded, placebo-controlled (Week 24)

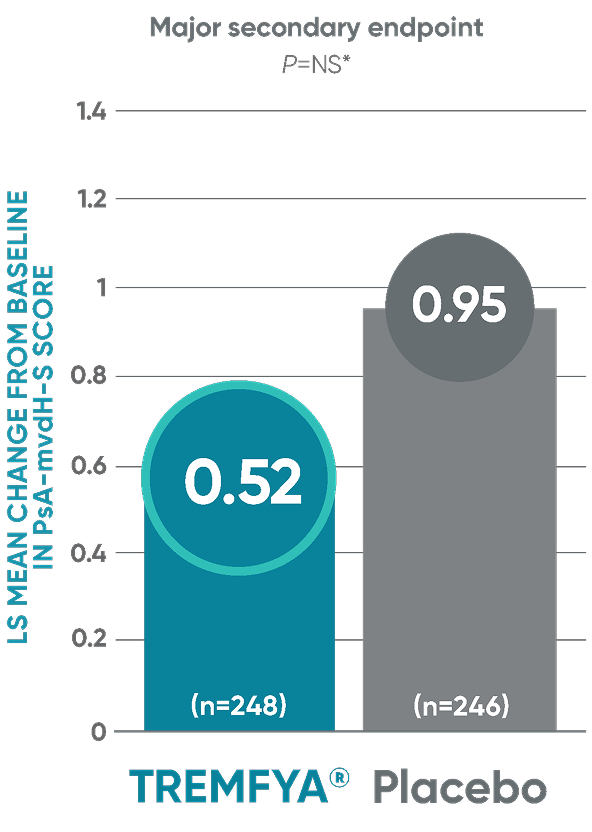
The results are not statistically significant; therefore, treatment effect for inhibition of structural damage has not been established.
Patients received TREMFYA® 100 mg SC at Week 0, Week 4, and every 8 weeks thereafter.
Treatment failure rules were not applied, and missing data were assumed to be missing at random and were imputed using
multiple imputation.
LS=least squares; mvdH-S=modified van der Heijde-Sharp; NS=not significant; SC=subcutaneous.
*P value is not significant.
IN ADULT PATIENTS WITH ACTIVE PsA
Durable* joint improvement with TREMFYA® at ~1 year,† consistent with pivotal trials1-3,6
APEX: ACR20/50/70 responses‡§
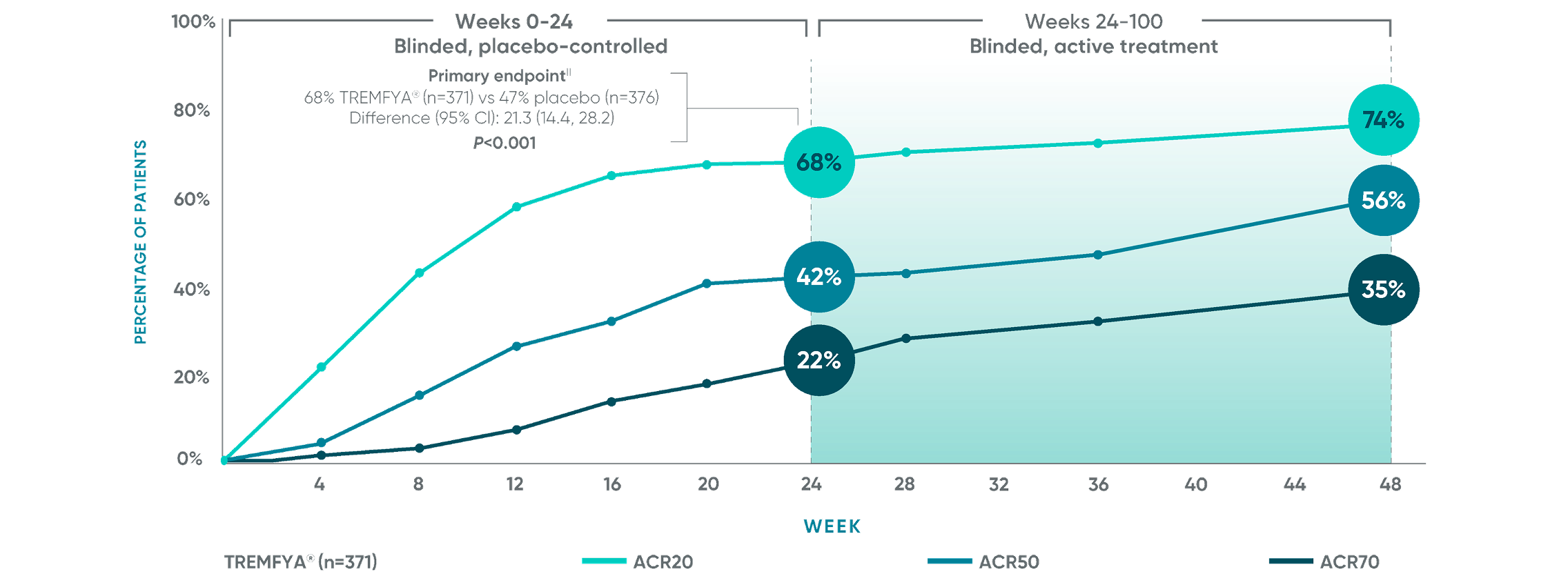
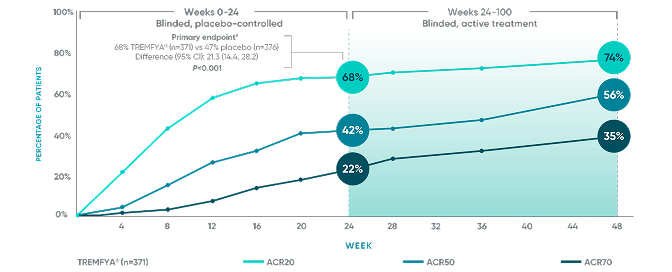
ACR50 and ACR70 responses at Week 24 were not adjusted for multiplicity; therefore, statistical significance has not been established. No statistical testing was conducted after Week 24 in the APEX trial.
Primary endpoint in pivotal studies: At Week 24, adult patients with active PsA receiving TREMFYA® demonstrated a greater clinical response
in ACR20 compared to placebo, in both the DISCOVER 1 (52% vs 22%) and DISCOVER 2 (64% vs 33%) trials, respectively (P<0.0001).7
ACR20=20% improvement in American College of Rheumatology composite measures of arthritis; ACR50=50% improvement in American College
of Rheumatology composite measures of arthritis; ACR70=70% improvement in American College of Rheumatology composite measures of arthritis;
CI=confidence interval; CMH=Cochran-Mantel-Haenszel; DMARD=disease-modifying antirheumatic drug; mFAS=modified full analysis set; MI=multiple imputation; ND/MD=natural disaster/major disruption; NRI=nonresponder imputation.
*Durable represents improvements at ~1 year.
†~1 year represents Week 48.
‡Efficacy analyses are from the mFAS, which included all randomized patients, excluding those from Ukraine sites rendered unable to support key study operations due to major disruptions. Shown are the average proportion of mFAS patients with ACR20/50/70 response, over the 200 MI datasets.
§Through Week 48, patients who discontinued study intervention for any reason except ND/MD were treated as nonresponders. Through Week 24, patients who initiated/
increased dose of oral corticosteroid or nonbiologic DMARD or initiated protocol-prohibited therapies for PsA were treated as nonresponders; after Week 24, these patients were not treated as nonresponders. Data from patients who discontinued study intervention or with severe treatment noncompliance due to ND/MD were imputed using MI at all subsequent timepoints or at the next timepoint, respectively. Other missing data were imputed using NRI.
||The primary endpoint P value is multiplicity-controlled using a fixed sequence testing procedure and can be used to determine statistical significance. Statistics are based on CMH across multiply imputed datasets.
GIVE YOUR ADULT PATIENTS WITH ACTIVE PsA THE CHANCE FOR
Joint preservation with TREMFYA®1-3,6
APEX: Significant (2.5x) reduction in structural damage progression vs placebo at Week 24
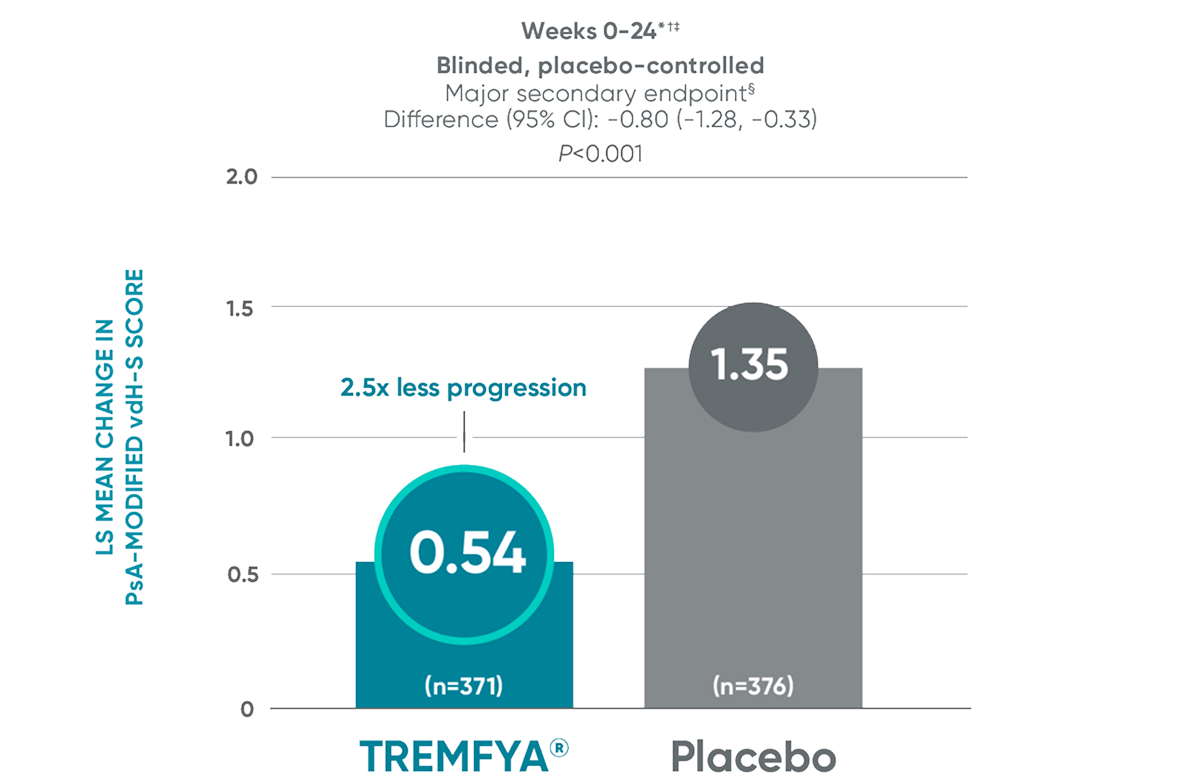
APEX: TREMFYA® shows sustained benefit at ~1 yearII
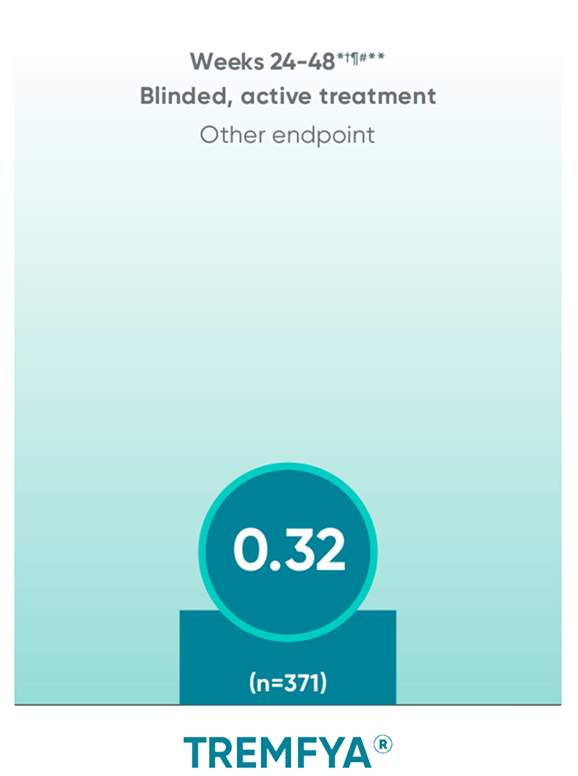
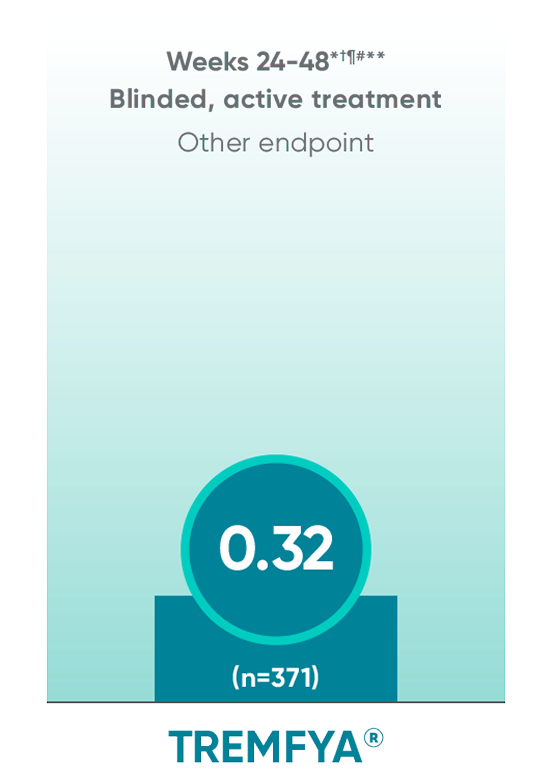
No statistical testing was conducted after Week 24 in the APEX trial.
ANCOVA=analysis of covariance; LS=least squares; vdH-S=van der Heijde-Sharp.
*Efficacy analyses are from the mFAS, which included all randomized patients, excluding those from Ukraine sites rendered unable to support key study operations due to major disruptions.
†Reading sessions 1 and 2 were prespecified in the protocol. Session 1 included independent assessment of images by two blinded central readers and a blinded adjudicator from Weeks 0 to 24. Session 2 included re-reading of images by the same two blinded central readers and a blinded adjudicator from Weeks 24 to 48.
‡For changes from baseline in total PsA-modified vdH-S score at Week 24, data from participants with discontinuation of study agent/severe noncompliance due to ND/MD were not used, and MI was employed for all missing data; treatment failure rules were not applied.
§The major secondary endpoint P value is multiplicity-controlled using a fixed sequence testing procedure and can be used to determine statistical significance. Statistics are based on ANCOVA across multiply imputed datasets.
||~1 year represents Week 48.
¶For patients with missing Week 48 radiographs, Week 0/24 radiographs were not re-read during reading session 2; Week 0/24 data from reading session 1 were used if available. The same readers were used for reading sessions 1 and 2.
#Score change was assessed in the mFAS according to the Adjusted Treatment Policy Estimand: irrespective of background PsA medication or adherence to study intervention; in situations when ND/MD occurred, observed data collected after ND/MD were not used; data for visits after ND/MD and missing data were imputed using full conditional specifications MI.
**LS mean change and CI were based on combining ANCOVA model (explanatory variables: baseline PsA-modified vdH-S score, treatment group, and randomization stratification level) results from each MI dataset.
IN ADULT PATIENTS WITH ACTIVE PsA
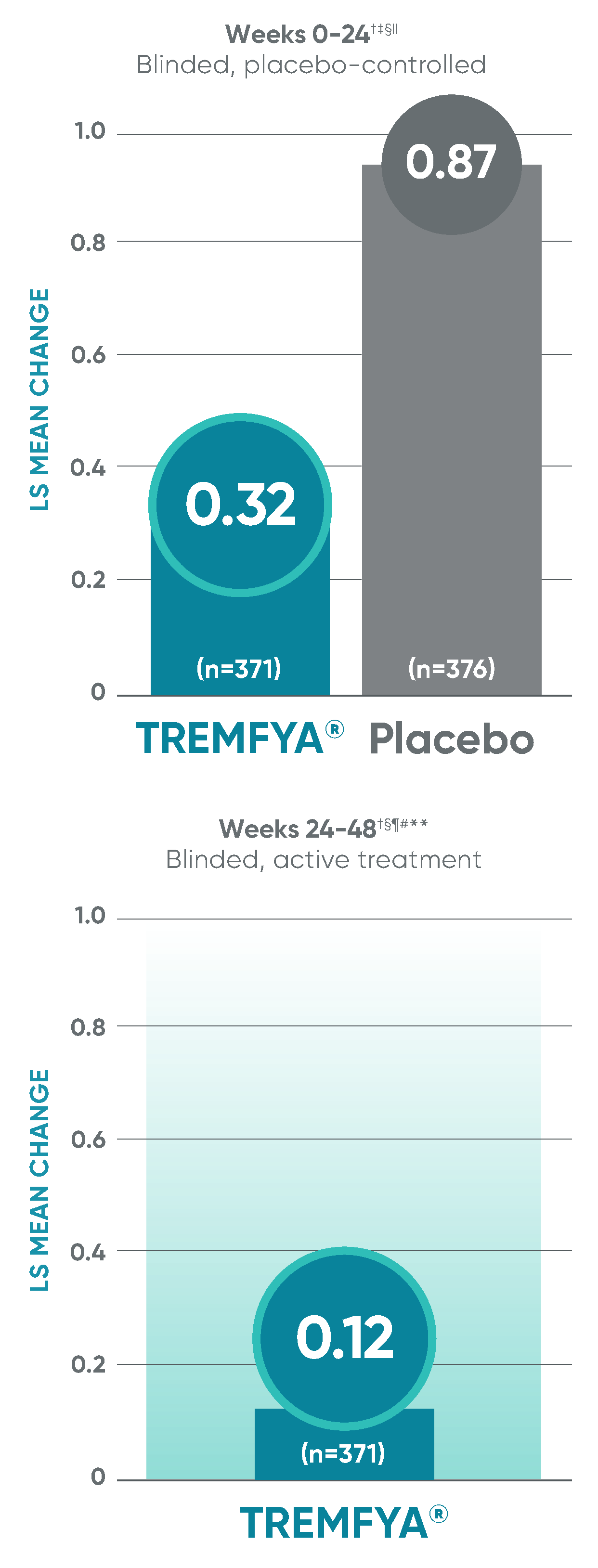
APEX: Change in joint erosion and joint space narrowing scores with TREMFYA® at Week 24 and ~1 year1-3,8*
Other endpoint:
Change in erosion score†

Other endpoint:
Change in JSN score†

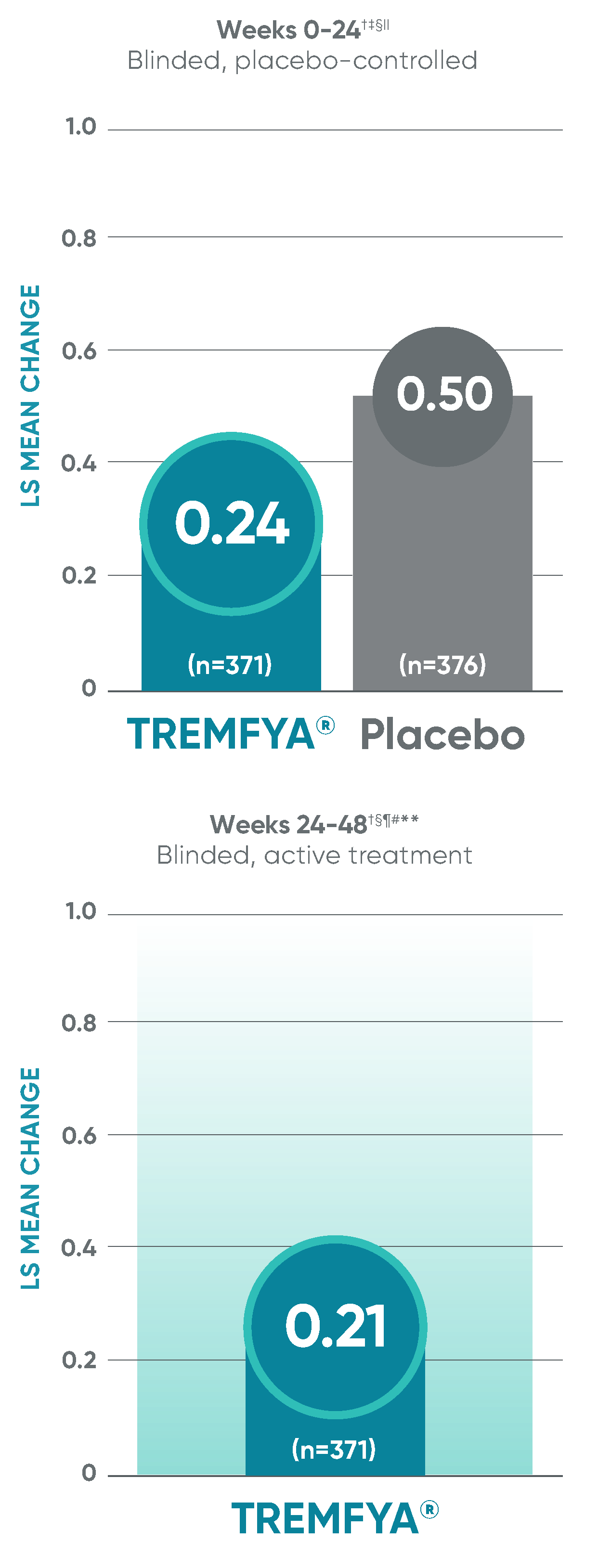
Erosion score and JSN score at Week 24 were not adjusted for multiplicity; therefore, statistical significance has not been established. No statistical testing was conducted after Week 24 in the APEX trial.
JSN=joint space narrowing.
*~1 year represents Week 48.
†Efficacy analyses are from the mFAS, which included all randomized patients, excluding those from Ukraine sites rendered unable to support key study operations due to major disruptions.
‡Statistics are based on ANCOVA across multiply imputed datasets.
§Reading sessions 1 and 2 were prespecified in the protocol. Session 1 included independent assessment of images by two blinded central readers and a blinded adjudicator from Weeks 0 to 24. Session 2 included re-reading of images by the same two blinded central readers and a blinded adjudicator from Weeks 24 to 48.
IIFor changes from baseline in erosion or JSN subscores at Week 24, data from participants with discontinuation of study agent/severe noncompliance due to ND/MD were not used, and MI was employed for all missing data; treatment failure rules were not applied.
¶Missing Data Imputation: After applying Intercurrent Event Strategies, the remaining missing data were imputed using MI. The corresponding n includes all subjects with change data at Week 48 after applying the Intercurrent Event
Strategies and Missing Data Imputation.
#Analysis Model: after applying the Intercurrent Event Strategy and Missing Data Imputation, data at Week 48 were analyzed using an ANCOVA model for each MI dataset. The explanatory variables of the ANCOVA model included baseline PsA-modified vdH-S score, treatment group, and the randomization stratification level.
**The LS Means and CI were based on combining ANCOVA model (explanatory variables: baseline PsA-modified vdH-S score, treatment group, and randomization stratification level) results from each MI dataset. The corresponding n includes all subjects with change data at that visit after applying the Intercurrent Event Strategy and Missing Data Imputation.
Erosion score and JSN score at Week 24 were not adjusted for multiplicity; therefore, statistical significance has not been established. No statistical testing was conducted after Week 24 in the APEX trial.
JSN=joint space narrowing.
*~1 year represents Week 48.
†Efficacy analyses are from the mFAS, which included all randomized patients excluding those from Ukraine sites rendered unable to support key study operations due to major disruptions.
‡Statistics are based on ANCOVA across multiply imputed datasets.
§Reading sessions 1 and 2 were prespecified in the protocol. Session 1 included independent assessment of images by two blinded central readers and a blinded adjudicator from Weeks 0 to 24. Session 2 included re-reading of images by the same two blinded central readers and a blinded adjudicator from Weeks 24 to 48.
IIFor changes from baseline in erosion or JSN subscores at Week 24, data from participants with discontinuation of study agent/severe noncompliance due to ND/MD were not used, and MI was employed for all missing data; treatment failure rules were not applied.
¶Missing Data Imputation: After applying Intercurrent Event Strategies, the remaining missing data were imputed using MI. The corresponding n includes all subjects with change data at Week 48 after applying the Intercurrent Event Strategies and Missing Data Imputation.
#Analysis Model: after applying the Intercurrent Event Strategy and Missing Data Imputation, data at Week 48 were analyzed using an ANCOVA model for each MI dataset. The explanatory variables of the ANCOVA model included baseline PsA-modified vdH-S score, treatment group, and the randomization stratification level.
**The LS Means and CI were based on combining ANCOVA model (explanatory variables: baseline PsA-modified vdH-S score, treatment group, and randomization stratification level) results from each MI dataset. The corresponding n includes all subjects with change data at that visit after applying the Intercurrent Event Strategy and Missing Data Imputation.
APEX: Proven safety profile through
Week 241-3
Adverse events reported in the placebo-controlled phase through Week 24 in APEX
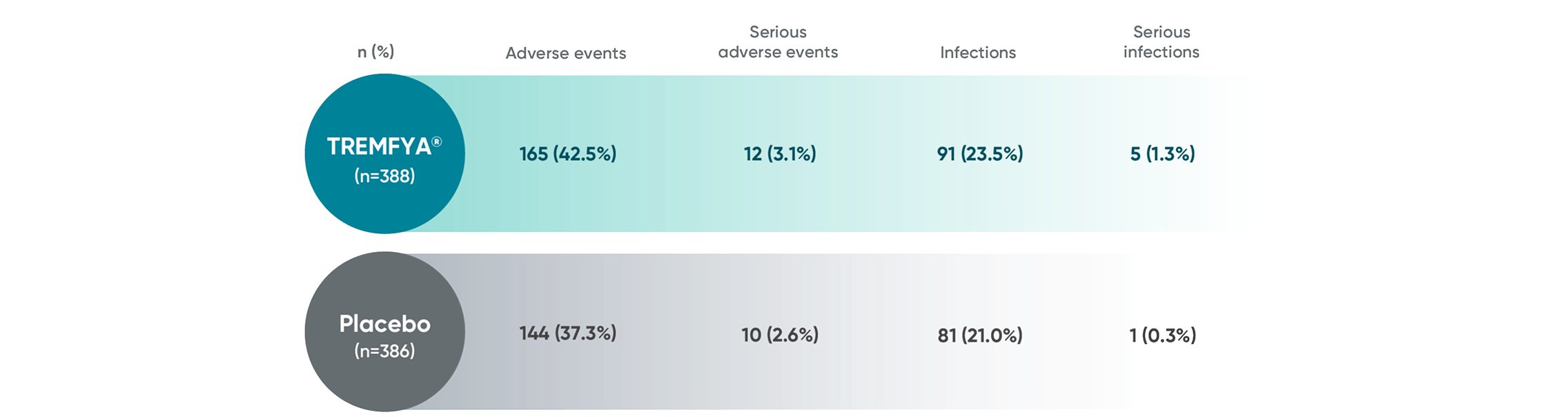
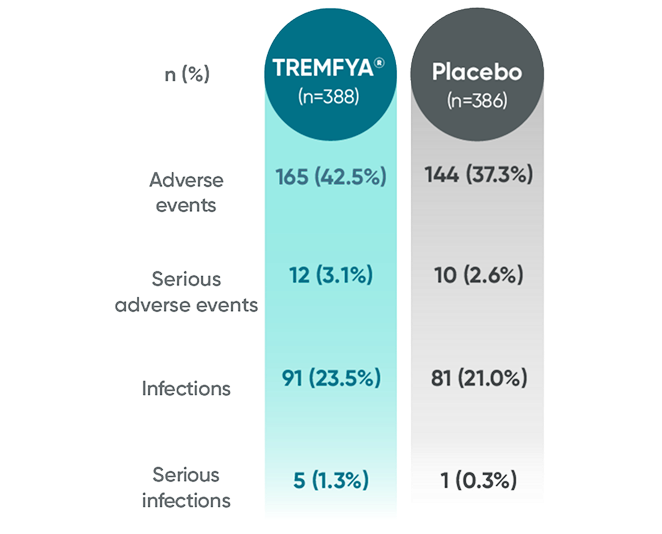
No new safety signals were reported through ~1 year1,3,6*
*~1 year represents Week 48.
References: 1. Mease PJ, Ritchlin CT, Coates LC, et al. Inhibition of structural damage progression with guselkumab, a selective IL-23i, in participants with active PsA: results through week 24 of the phase 3b, randomized, double-blind, placebo-controlled APEX study. Oral presentation at: European Alliance of Associations for Rheumatology (EULAR) 2025 Congress; June 11-14, 2025; Barcelona, Spain. 2. Mease PJ, Ritchlin CT, Coates LC, et al. Inhibition of structural damage progression with guselkumab, a selective IL-23i, in participants with active PsA: results through week 24 of the phase 3b, randomized, double-blind, placebo-controlled APEX study. Abstract presented at: European Alliance of Associations for Rheumatology (EULAR) 2025 Congress; June 11-14, 2025; Barcelona, Spain. Late-Breaking Abstracts Session II.
3. Mease PJ, Ritchlin CT, Coates LC, et al. Inhibition of structural damage progression with the selective interleukin-23 inhibitor guselkumab in participants with active PsA: results through week 24 of the phase 3b, randomised, double-blind, placebo-controlled APEX study. Ann Rheum Dis. 2025;84(12):1983-1994. 4. Mease PJ, Rahman P, Gottlieb AB, et al. Guselkumab in biologic-naïve patients with active psoriatic arthritis (DISCOVER-2): a double-blind, randomised, placebo-controlled phase 3 trial. Lancet. 2020;395(10230):1126-1136. 5. Ritchlin CT, Coates LC, Mease PJ, et al. The effect of guselkumab on inhibiting radiographic progression in patients with active psoriatic arthritis: study protocol for APEX, a Phase 3b, multicenter, randomized, double-blind, placebo-controlled trial. Trials. 2023;24(1):22. 6. Ritchlin CT, Mease PJ, Coates LC, et al. Durable inhibition of structural damage progression and improvements in joint disease activity with guselkumab in active and erosive psoriatic arthritis: Week 48 results from APEX. Presented at: 6th Inflammatory Skin Disease Summit (ISDS); November 12-15, 2025; New York, NY. 7. TREMFYA® (guselkumab) [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc. 8. Data on file. Janssen Biotech, Inc.


