For US Healthcare Professionals
I am a:


Joint improvement proven to last at 2 years1-5*†‡§II¶#
DISCOVER 2 ACR20/50/70: Open-label active treatment, NRI post hoc analysis (Weeks 24-100)


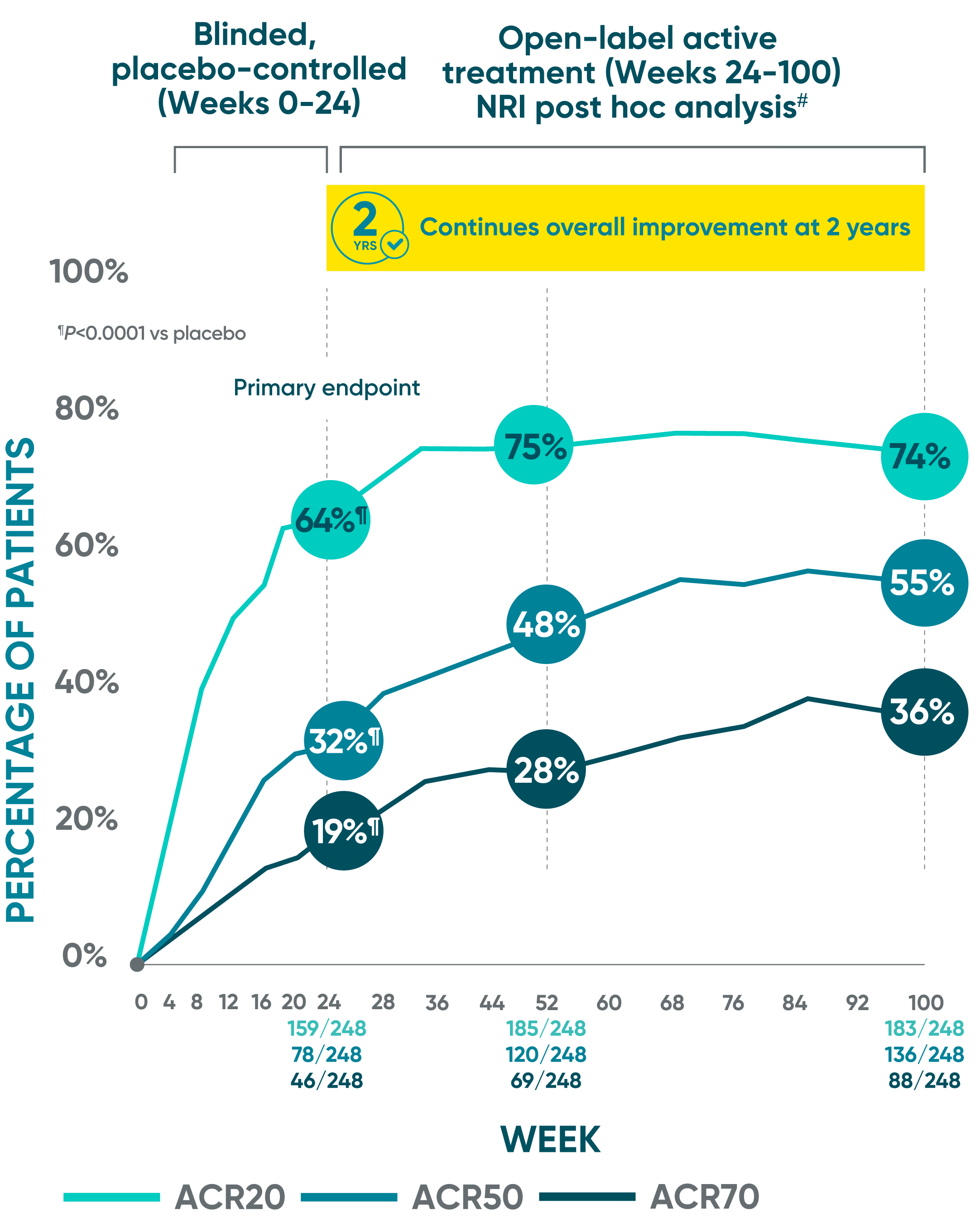
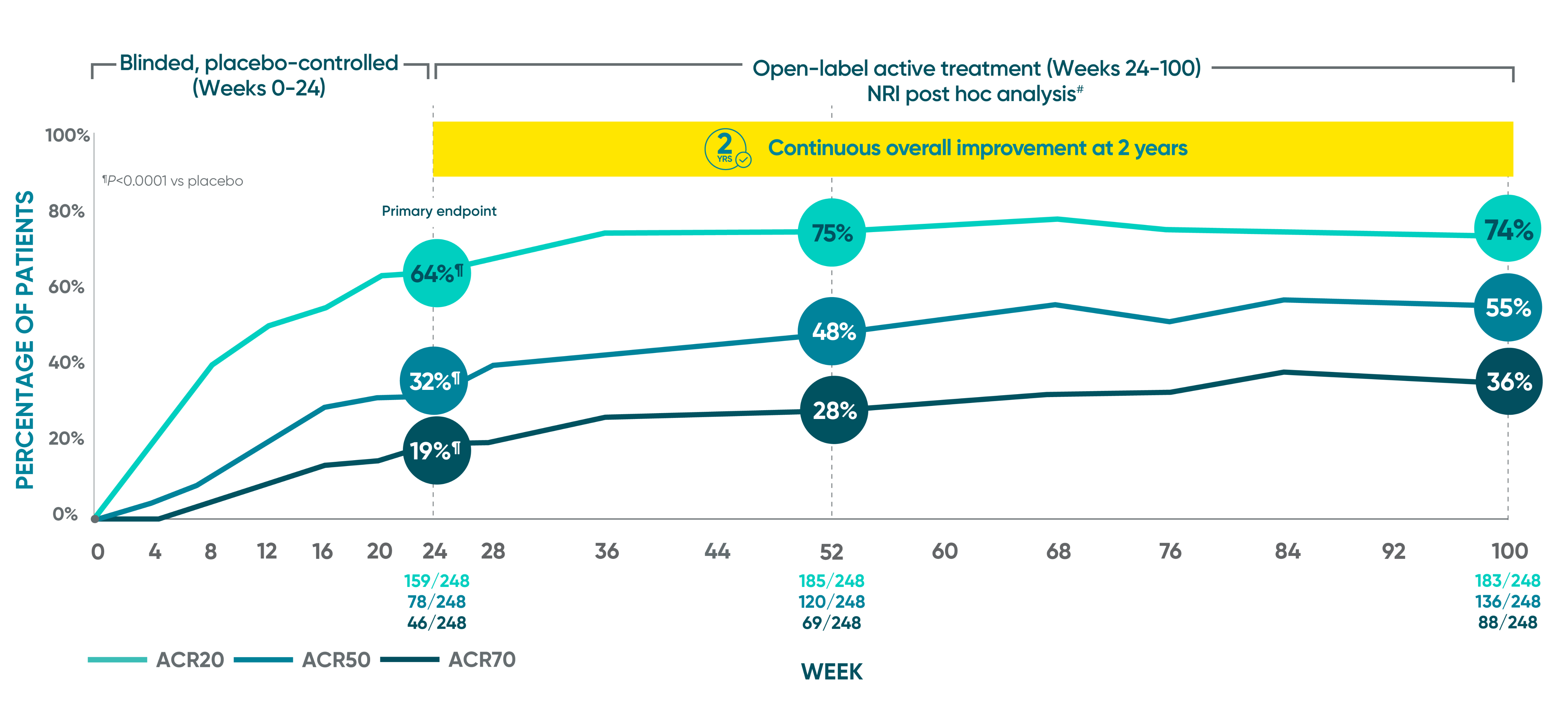
ACR50 and ACR70 at Week 24 were not part of the sequential testing procedure but were prespecified to be tested upon achieving statistical significance for ACR20 at Week 24.
DISCOVER 2 Week 24: Blinded, placebo-controlled NRI¶
ACR20 placebo response: 33% (81/246)
ACR50 placebo response: 14% (35/246)
ACR70 placebo response: 4% (10/246)

Prescribed** IL-23i in active PsA by rheumatologists
**#1 prescribed refers to year-to-year date TREMFYA® active PsA market share vs IL-23 inhibitors as of December 2024.
IL-23i=interleukin-23 inhibitor
Source: IQVIA Weekly LAAD as of 12/13/24 X Free Goods; Total PsA EqU share for Rheumatologists.
NRI=nonresponder imputation.
*The same patients may not have responded at each time point.
†Year 2 represents Week 100.
‡Through Week 24, patients were considered to be nonresponders after meeting treatment failure criteria (see study designs). After Week 24, treatment failure rules were not applied.
§Patients with missing data were considered nonresponders.
||After Week 24, patients and doctors knew that all patients were on TREMFYA® (open label with a blinded dosing interval), which may have affected the results.
#The DISCOVER 2 prespecified as-observed analysis from Weeks 24 to 100 is not shown.
References: 1. TREMFYA® (guselkumab) [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc. 2. Data on file. Janssen Biotech, Inc. 3. Mease PJ, Rahman P, Gottlieb AB, et al. Guselkumab in biologic-naïve patients with active psoriatic arthritis (DISCOVER-2): a double-blind, randomised, placebo-controlled phase 3 trial. Lancet. 2020;395(10230):1126-1136. 4. McInnes IB, Rahman P, Gottlieb AB, et al. Efficacy and safety of guselkumab, a monoclonal antibody specific to the p-19 subunit of interleukin-23, through two years: results from a phase III, randomized, double-blind, placebo-controlled study conducted in biologic-naïve patients with active psoriatic arthritis. Arthritis Rheumatol. 2022;74(3):475-485. 5. McInnes IB, Rahman P, Gottlieb AB, et al. Efficacy and safety of guselkumab, an interleukin-23p19-specific monoclonal antibody, through one year in biologic-naïve patients with psoriatic arthritis. Arthritis Rheumatol. 2021;73(4):604-616.
DISCOVER 1: ACR20/50/70
Open-label active treatment, NRI post hoc analysis (Weeks 24-52)1-4


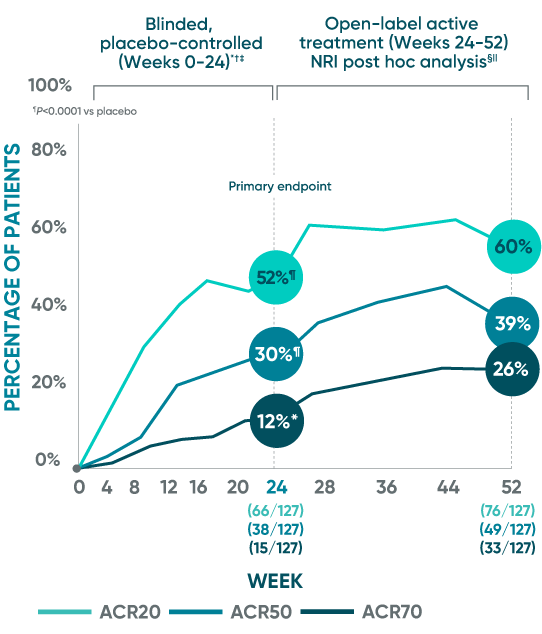
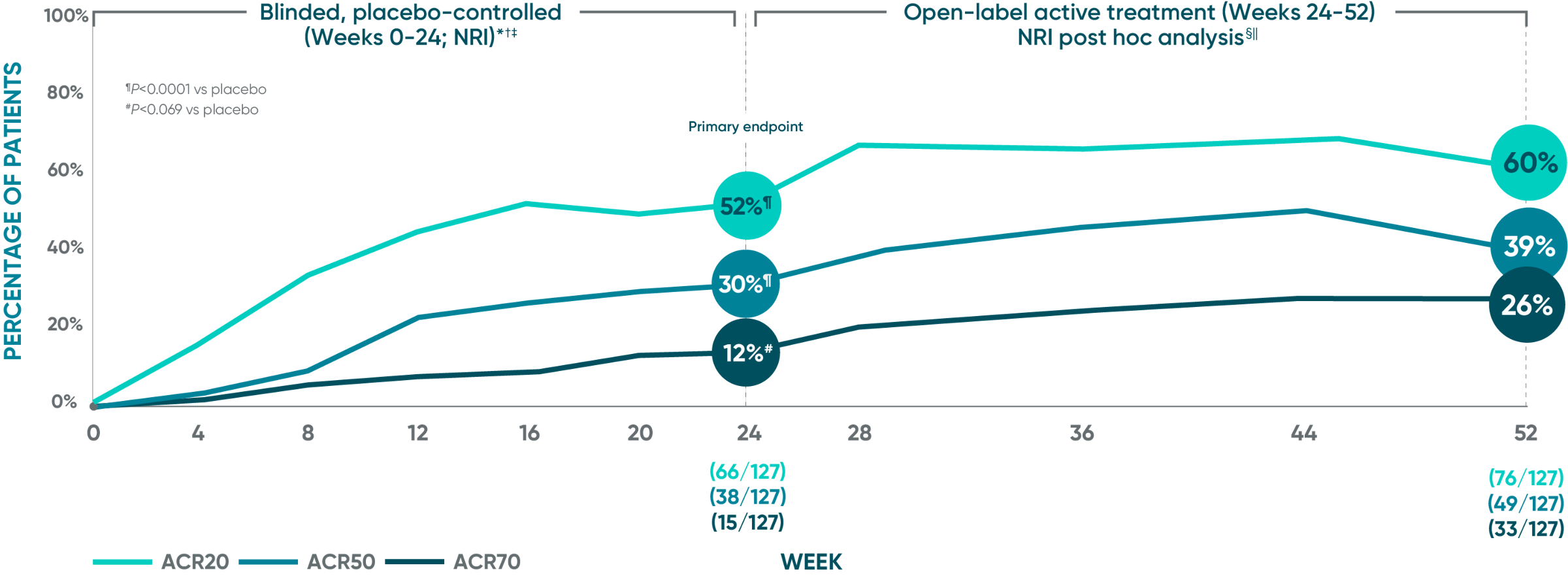
ACR50 and ACR70 response at Week 24 was not part of the sequential testing procedure but was prespecified to be tested upon achieving statistical significance for ACR20 at Week 24.
DISCOVER 1 Week 24: Blinded, placebo-controlled (NRI)*†‡
Week 24: Blinded, placebo-controlled (NRI)*†‡
ACR20 response (primary endpoint): 52% TREMFYA® (66/127) vs 22% placebo (28/126) (P<0.0001)
ACR50 response (secondary endpoint): 30% TREMFYA® (38/127) vs 9% placebo (11/126) (P<0.0001)
ACR70 response (secondary endpoint): 12% TREMFYA® (15/127) vs 6% placebo (7/126) (P=0.069)
NRI=nonresponder imputation.
*The same patients may not have responded at each time point.
†Through Week 24, patients were considered to be nonresponders after meeting treatment failure criteria (see study designs). After Week 24, treatment failure rules were not applied.
‡Patients with missing data were considered nonresponders.
§After Week 24, patients and doctors knew that all patients were on TREMFYA® (open label with a blinded dosing interval), which may have affected the results.
||The DISCOVER 1 prespecified as-observed analysis from Week 24 to Week 52 is not shown.
References: 1. TREMFYA® (guselkumab) [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc. 2. Data on file. Janssen Biotech, Inc. 3. Deodhar A, Helliwell PS, Boehncke W-H, et al. Guselkumab in patients with active psoriatic arthritis who were biologic-naïve or had previously received TNFα inhibitor treatment (DISCOVER 1): a double-blind, randomised, placebo-controlled phase 3 trial. Lancet. 2020;395(10230):1115-1125. 4. Ritchlin CT, Helliwell PS, Boehncke W-H, et al. Guselkumab, an inhibitor of the IL-23p19 subunit, provides sustained improvement in signs and symptoms of active psoriatic arthritis: 1 year results of a phase III randomised study of patients who were biologic-naïve or TNFα inhibitor-experienced. RMD Open. 2021; 7(1): e001457.
FOR ADULT PATIENTS WITH ACTIVE PsA
Complete resolution of enthesitis and dactylitis at 2 years
with TREMFYA®1-6*†‡§II¶#
Pooled data from DISCOVER 1 and DISCOVER 2 at Week 24 and 52 and from only DISCOVER 2 at Week 100


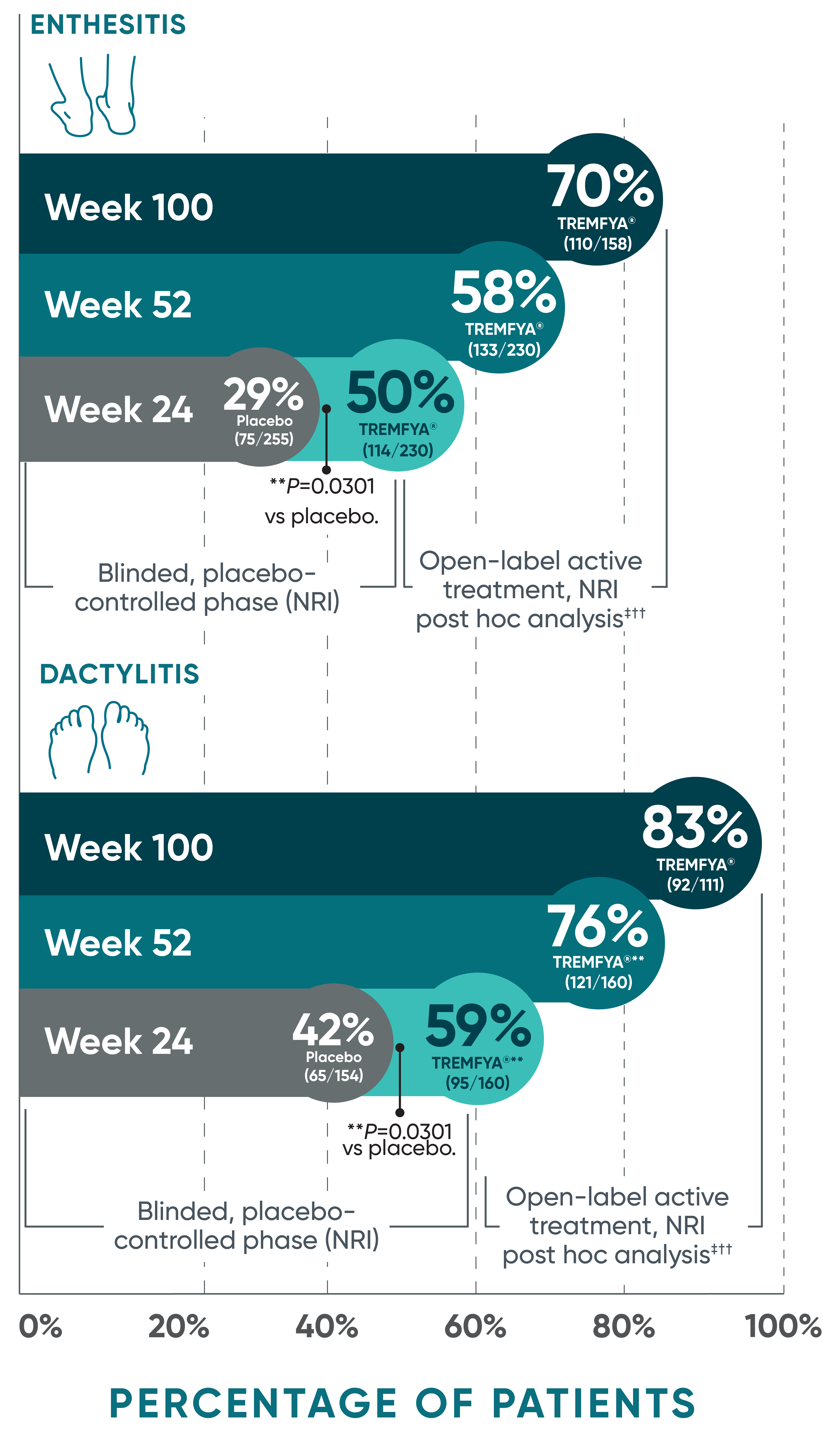
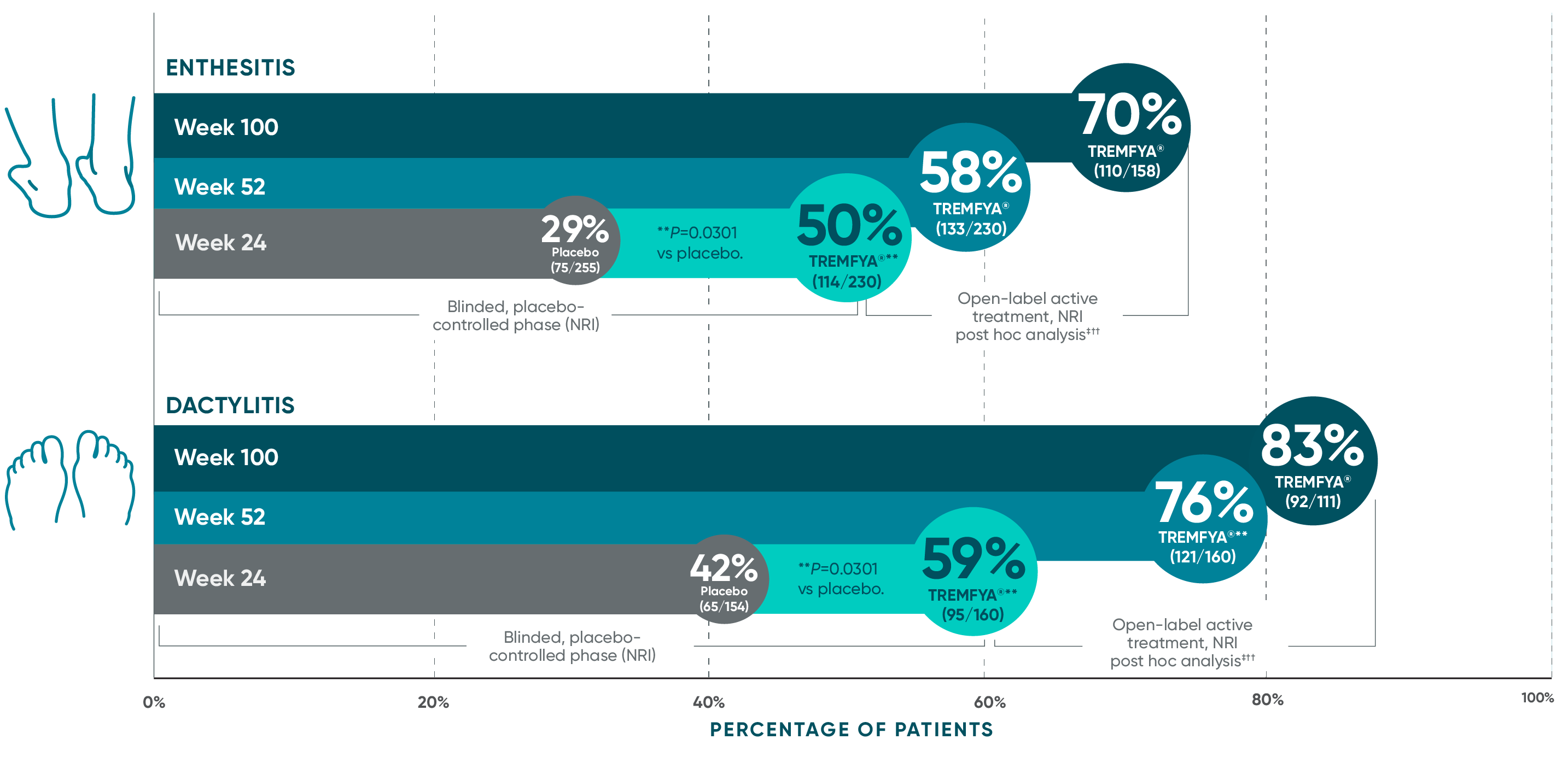
Complete resolution of enthesitis and dactylitis are defined as LEI score=0 and dactylitis score=0, respectively.
LEI=Leeds Enthesitis Index; NRI=nonresponder imputation.
*The same patients may not have responded at each time point.
†Year 2 represents Week 100.
‡After Week 24, patients and doctors knew that all patients were on TREMFYA® (open label with a blinded dosing interval), which may have affected the results.
§Among patients with LEI enthesitis score >0 at baseline.
||Among patients with dactylitis score >0 at baseline.
¶Through Week 24, patients were considered to be nonresponders after meeting treatment failure criteria (see study designs). After Week 24, treatment failure rules were not applied.
#Patients with missing data were considered nonresponders.
††The prespecified as-observed analyses at Week 52 and Week 100 are not shown.
References: 1. TREMFYA® (guselkumab) [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc. 2. Data on file. Janssen Biotech, Inc. 3. Mease PJ, Rahman P, Gottlieb AB, et al. Guselkumab in biologic-naïve patients with active psoriatic arthritis (DISCOVER-2): a double-blind, randomised, placebo-controlled phase 3 trial. Lancet. 2020;395(10230):1126-1136.
4. McInnes IB, Rahman P, Gottlieb AB, et al. Efficacy and safety of guselkumab, a monoclonal antibody specific to the p-19 subunit of interleukin-23, through two years: results from a phase III, randomized, double-blind, placebo-controlled study conducted in biologic-naïve patients with active psoriatic arthritis. Arthritis Rheumatol. 2022;74(3):475-485. 5. McInnes IB, Rahman P, Gottlieb AB, et al. Efficacy and safety of guselkumab, an interleukin-23p19-specific monoclonal antibody, through one year in biologic-naïve patients with psoriatic arthritis. Arthritis Rheumatol. 2021;73(4):604-616. 6. McInnes IB, Rahman P, Gottlieb AB, et al. Efficacy and safety of guselkumab, a monoclonal antibody specific to the p-19 subunit of interleukin-23, through 2 years: results from a phase 3, randomised, double-blind, placebo-controlled study conducted in biologic-naive patients with active psoriatic arthritis. Presented at: Innovations in Dermatology 2021; Virtual; March 16-20, 2021.
IN ADULTS WITH MODERATE TO SEVERE PLAQUE PsO
