For US Healthcare Professionals
I am a:


PROTOSTAR Study: TREMFYA® skin clearance rates in pediatric patients at Week 161,2
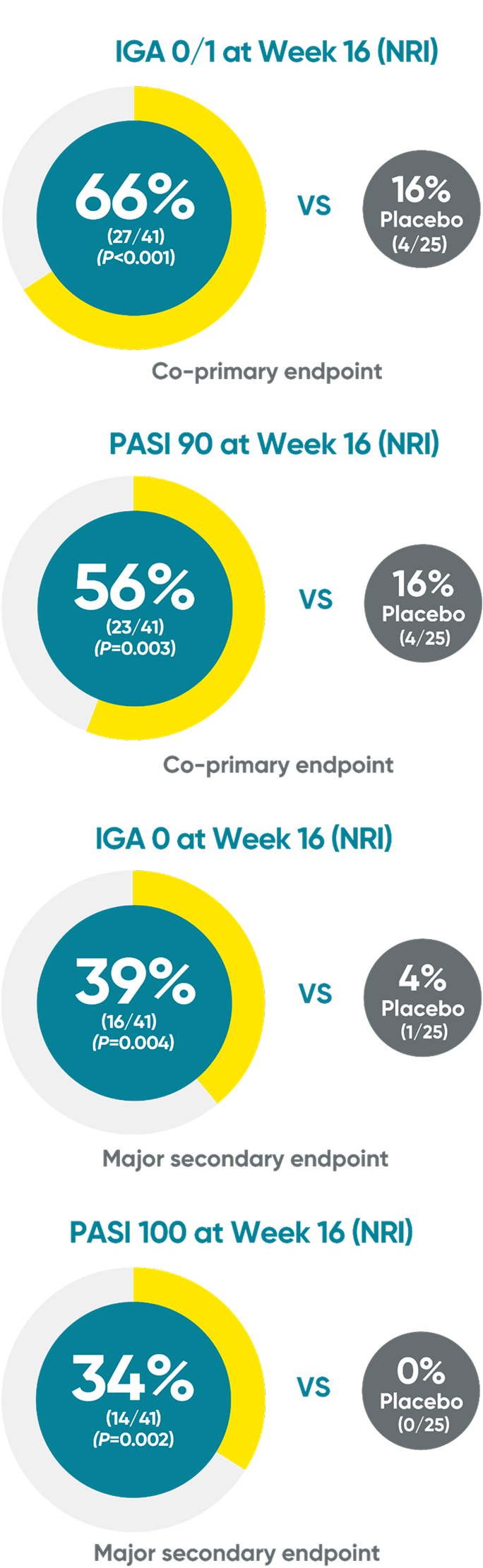
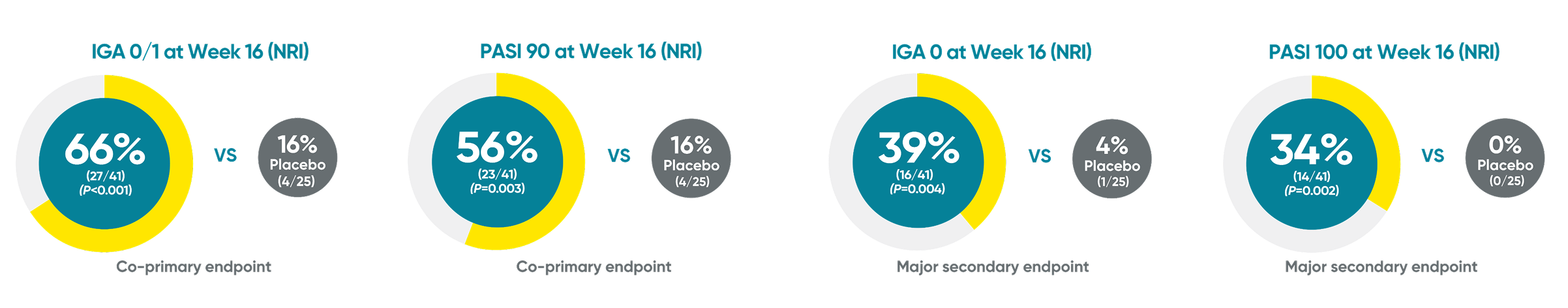
Nonresponder imputation (NRI) was used for analyses.
IGA=Investigator’s Global Assessment; PASI=Psoriasis Area and Severity Index.
References: 1. TREMFYA® (guselkumab) [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc. 2. Prajapati V, Seyger M, Wilsmann-Theis D, et al. Guselkumab for the treatment of moderate-to-severe plaque psoriasis in paediatric patients: results of the phase III randomized placebo-controlled PROTOSTAR study. Br J Dermatol. 2025;192:618-628.
IN PEDIATRIC PATIENTS 6 YEARS OF AGE AND OLDER WHO ALSO WEIGH AT LEAST 40 KG WITH MODERATE TO SEVERE PLAQUE PSORIASIS (PsO)
PROTOSTAR Safety Results2
The safety profile observed in pediatric patients 6 years of age and older treated with TREMFYA® up to 52 weeks was consistent with the safety profile observed in adult patients with moderate to severe plaque psoriasis.
PROTOSTAR: Adverse events reported at Week 16


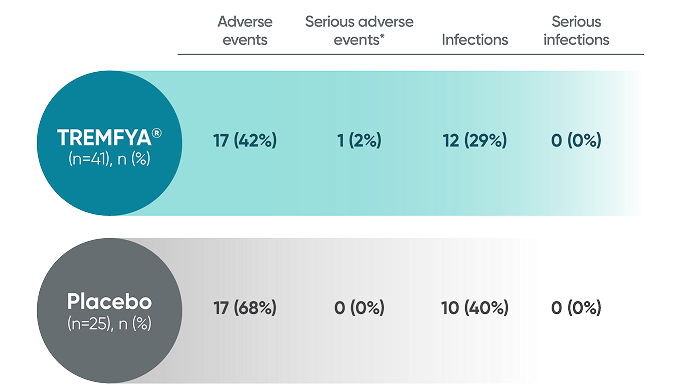
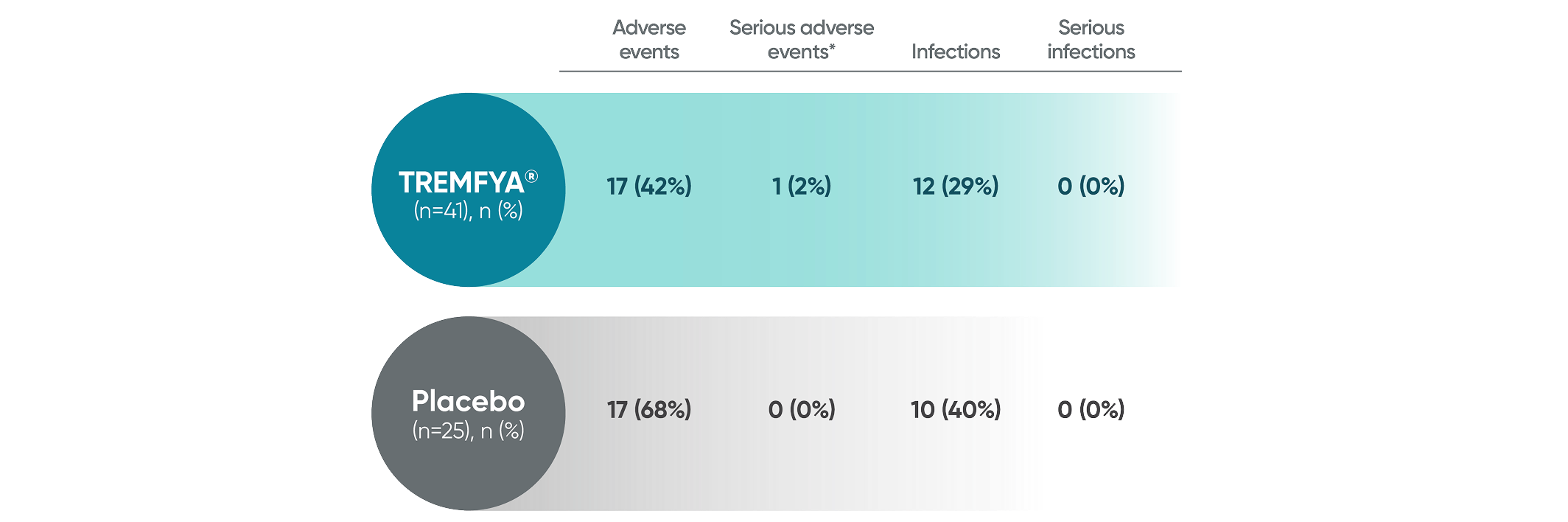
IL-23i=interleukin-23 inhibitor; PsO=psoriasis.
*Included a radius fracture (TREMFYA® group).
References: 1. TREMFYA® (guselkumab) [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc. 2. Prajapati V, Seyger M, Wilsmann-Theis D, et al. Guselkumab for the treatment of moderate-to-severe plaque psoriasis in paediatric patients: results of the phase III randomized placebo-controlled PROTOSTAR study. Br J Dermatol. 2025;192:618-628.
Pediatric Dosing
TREMFYA® is administered by subcutaneous injection at Week 0, Week 4, and every 8 weeks thereafter.
The recommended dose for adults and pediatric patients 6 years of age and older who also weigh at least 40 kg is 100 mg.
Pretreatment Evaluation: Evaluate for tuberculosis (TB) infection; if clinically indicated, evaluate liver enzymes and bilirubin levels, and complete all age-appropriate vaccinations according to current immunization guidelines.
Monitor: For signs and symptoms of active TB during and after treatment with TREMFYA®. If clinically indicated, evaluate liver enzymes and bilirubin levels periodically according to routine patient management.
TREMFYA® is intended for use under the guidance and supervision of a healthcare professional. After proper training in subcutaneous injection technique, adults may self-inject. Pediatric self-administration is not recommended. Administration of TREMFYA® to pediatric patients should be performed by a healthcare provider or by a caregiver who has received training and demonstrated proper subcutaneous injection technique.
Reference: 1. Prajapati V, Seyger M, Wilsmann-Theis D, et al. Guselkumab for the treatment of moderate-to-severe plaque psoriasis in paediatric patients: results of the phase III randomized placebo-controlled PROTOSTAR study. Br J Dermatol. 2025;192:618-628.
IN ADULT PATIENTS WITH MODERATE TO SEVERE PLAQUE PsO
